The concentration of a solution is often expressed as the percentage of solute in the total amount of solution. For the extremely dilute solutions the concentration unit parts per million (ppm) is often used. Since the amounts of solute and solution present can be stated in terms of either mass or volume, different types of percent and ppm exist:
This module is to compute the mass-volume percentage of solute and parts per million (ppm) or to calculate solute or solvent by knowing concentration. Notice that the units shall be either gram/mL or kg/L.
(m/v) % = [mass of solute (g) / volume of solution (mL)] x 100
ppm (m/v) = [mass of solute (g) / volume of solution (mL)] x 106
1. Calculate concentration by inputting the solute and solution amounts in the above formulae:
concentration = solute / solution
2. Knowing the concentration and one of two: solute or solution, calculate the other one:
solute = concentration x solution
solution = solute / concentration
This is one step process, enter the known data and press Calculate to output the unknowns.
1. Select Mass-Volume Percent link from the front page or Mass-Volume Percent tab from the Solution module. The Input and Output screen appears.
2. In the Input area, enter the two known quantities with a proper significant figure. Select the units associated with the input.
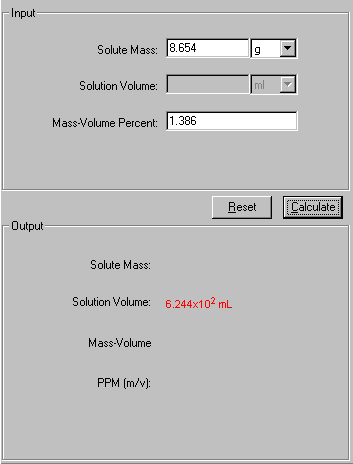
3. Click Calculate to output the answer.
4. The Show Work area on the right shows you step-by-step how your problem has been solved.
To start a new problem, click Reset. All Input fields will be cleared. Follow Step 1-3 again.