Weak Acid Dissociation
Weak acids can dissociate in an aqueous solution to achieve equilibrium. The percent dissociation is defined as follows:
Percent dissociation = amount dissociated (mol/L) / initial concentration (mol/L) x 100%
For example,
HC2H3O2 (aq) = H+ (aq) + C2H3O2- (aq)
The equilibrium expression is
Ka = 1.8 x 10-5 = [H+][C2H3O2-] / [HC2H3O2]
The percent dissociation can be calculated as:
Percent dissociation = [H+] (final) / [HC2H3O2] (initial)
This module can perform the following calculations:
1. Known initial concentration of a weak acid and Ka, calculate percent dissociation and pH.
2. Known initial concentration of a weak acid and percent dissociation, calculate Ka and pH.
3. Known initial concentration of a weak base and Kb, calculate pH.
Example,
For acetic acid with the initial concentration of 1.00M, Ka = 1.8 x 10-5,calculate % dissociation and pH.
This is one step process, enter the known data and press Calculate to output the unknowns.
1. Select Weak Acid Dissociation link from the front page or Weak Acid dissociation tab from the Acid, Baseand Salt module. The Input and Output screen appears.
2. In the Input area, enter the known quantities with a proper significant figure.
3. Click Calculate to output the answer.
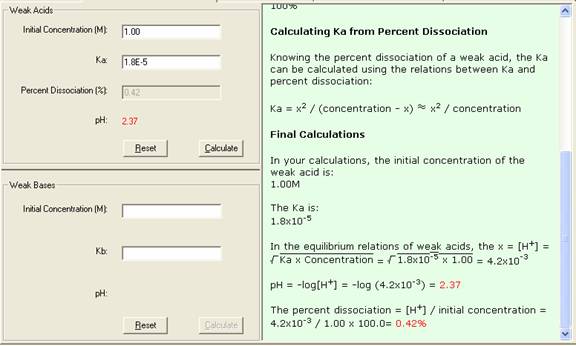
4. The Show Work area on the right shows you step-by-step how your problem has been solved.
To start a new problem, click Reset. All Input fields will be cleared. Follow Step 1-3 again.